Why Are FDA Approved Orthopedic Implants Essential for Successful Surgery?
The success of orthopedic surgeries largely depends on the quality of the materials used. FDA approved orthopedic implants play a crucial role in this context. These implants undergo rigorous testing and evaluation to ensure their safety and effectiveness. This process helps provide surgeons and patients with confidence in their choices.
Orthopedic implants must withstand the stresses of daily life. Without FDA approval, there is no guarantee of reliability. Some patients may experience complications from non-approved alternatives. This can lead to prolonged recovery times and additional surgeries. A deep understanding of FDA regulations can aid in making informed decisions.
While FDA approved orthopedic implants offer many benefits, challenges still exist. Not every patient responds the same way to these implants. Variability in healing processes can make outcomes unpredictable. Therefore, continual evaluation of implant performance is essential in the ever-evolving field of orthopedic surgery.

Importance of FDA Approval in Orthopedic Implants
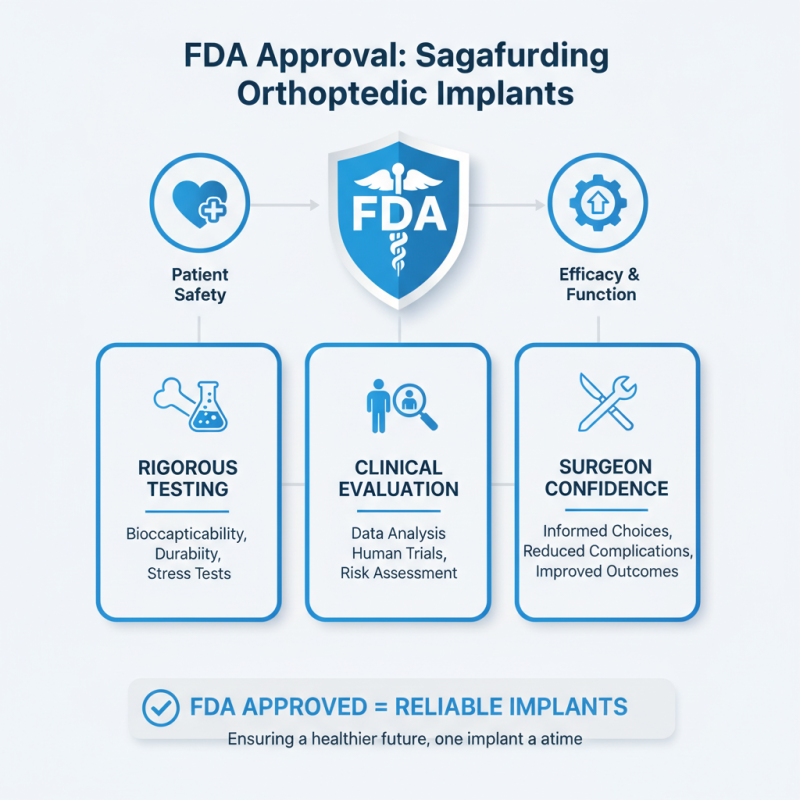
FDA approval plays a vital role in ensuring the safety and efficacy of orthopedic implants. These devices must go through rigorous testing and evaluation. The process guarantees that the implants can withstand stress and function correctly in the human body. Surgeons rely on these approvals to make informed decisions for their patients. Choosing FDA-approved implants can significantly reduce the risk of complications during surgery.
When selecting an orthopedic implant, understanding the approval process is important. Ask your surgeon about the implant's history. Inquire about any potential risks. This knowledge helps in making a wise decision.
Tip: Always consult your physician before undergoing surgery. They can explain the intricacies of the implants used.
The landscape of orthopedic surgery is constantly evolving. New technologies and materials are available. However, not all of them undergo thorough scrutiny. Some options might be less tested, raising questions about their reliability. It’s essential to balance innovation with proven safety.
Tip: Stay updated with new studies on orthopedic implants. Research trends can impact clinical decisions.
Role of Quality Standards in Ensuring Patient Safety
Choosing FDA-approved orthopedic implants is crucial for surgical success. The FDA sets rigorous quality standards. These standards are designed to enhance patient safety and ensure effective results. When healthcare providers use these implants, they rely on years of research and testing. Each implant must undergo extensive evaluation before approval.
Quality standards help minimize the risks associated with surgery. They ensure that materials used are safe and effective. Implants must resist infection and function properly over time. However, lapses can occur. Some products may show failure after surgery. Continuous monitoring and evaluations are necessary to improve these standards.
Patients should be aware of the importance of quality. Discussing these aspects with surgeons can foster trust. Open communication empowers patients to make informed decisions. Increased transparency around these standards can enhance patient safety overall. Understanding the complexities in implant approvals encourages a more robust healthcare system.
Impact of FDA Approval on Implant Performance and Longevity
FDA approval plays a crucial role in the performance and longevity of orthopedic implants. It involves rigorous testing and evaluation. These processes ensure that the implants are safe and effective for patients. Approved devices undergo trials that assess not only their effectiveness but also their long-term stability within the body. This assures healthcare providers and patients of their reliability during surgery and recovery.
Tips for patients: Always ask your surgeon about the implants being used. Understanding the approval status can enhance your confidence in the procedure. Additionally, seek information on similar past surgeries. This can provide insight into potential outcomes and any complications faced.
Moreover, FDA-approved implants may still have limitations. Variability in patient anatomy can affect how well an implant performs. Different individuals may respond differently to the same device. This emphasizes the importance of personalized medical advice. Always discuss your unique health conditions with your medical team for the best possible results.
Consequences of Using Non-FDA Approved Orthopedic Implants
Using non-FDA approved orthopedic implants can lead to severe consequences. These implants may not have undergone exhaustive testing for safety and effectiveness. Without this assurance, patients risk complications during and after surgery.
Pain, infection, and implant failure are potential outcomes. These issues may require additional surgeries to address.
Patients deserve transparency about the implants they receive. Non-FDA approved devices may be cheaper, but the risks can outweigh the savings. Surgeons often adhere to standard practices, emphasizing the use of reliable products.
Non-compliance with FDA regulations could signal underlying problems with quality or manufacturing. This creates a dilemma for healthcare providers and patients alike.
Moreover, the lack of accountability associated with unapproved implants raises concerns. Manufacturers may not have a track record of quality assurance. Healthcare professionals must remain vigilant. They should discuss the implications of using unverified products with patients.
Choosing FDA approved implants fosters trust and ensures a commitment to patient safety. It is essential to prioritize health over cost in orthopedic surgery.
Future Trends in FDA Regulations for Orthopedic Devices
The future of FDA regulations for orthopedic devices is evolving. As technology advances, standards must adapt. Personalized medicine is becoming more prevalent, pushing the FDA to adjust its approach. Patients today expect more tailored solutions. This shift demands robust regulatory frameworks to ensure safety and efficacy.
One trend is the emphasis on real-world evidence. The FDA recognizes that clinical trials may not capture all potential device interactions. Gathering data from actual outcomes enhances our understanding of how these implants perform. However, this shift raises questions regarding data integrity and reliability. Stakeholders must address these concerns proactively.
Another focus is on digital health integration. Orthopedic devices are increasingly incorporating smart technology. This provides continuous monitoring and feedback. The FDA is assessing how to regulate these innovations effectively. Ensuring patient safety without stifling innovation poses a significant challenge. Balancing regulation and advancement requires careful thought and collaboration among experts.
Importance of FDA Approved Orthopedic Implants
Related Posts
-

What is Joint Replacement Mobility Restoration and How Does it Work?
-

Why Is Total Knee Replacement Care Essential for a Successful Recovery?
-
How to Find the Best Support for Hip Replacement Patients?
-
China Best Consensus Knee System Innovations and Benefits Explained?
-

Best Partial Knee Replacement Care Options Worldwide?
-

2026 How to Find Effective Hip Replacement Patient Support and Recovery Tips
Why Shalby MedTech
We are committed to deliver solutions and transforming the way joint replacement procedures are performed.


 Looking for the
Looking for the  Register as
Register as